Painstaking Lessons Of Info About How To Write Valence Electron Configuration

The elements that form bonds by donating electrons are called cation.
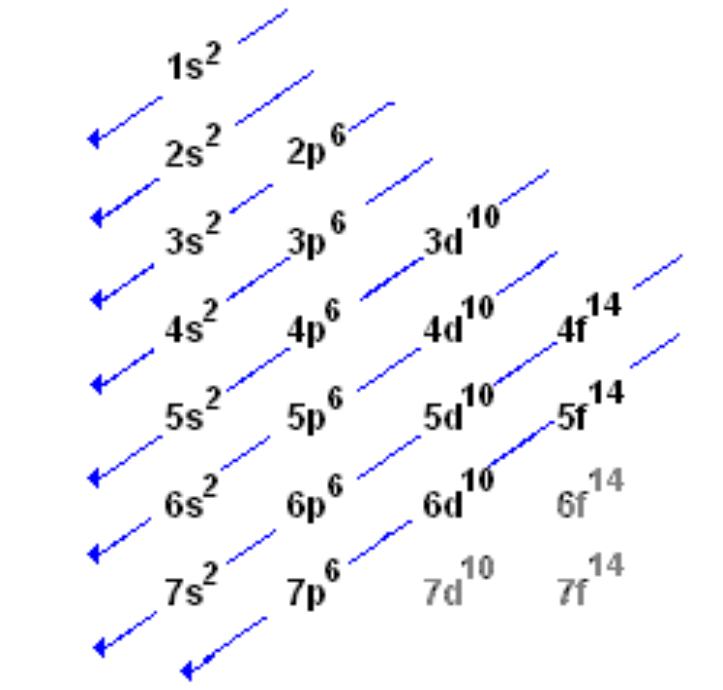
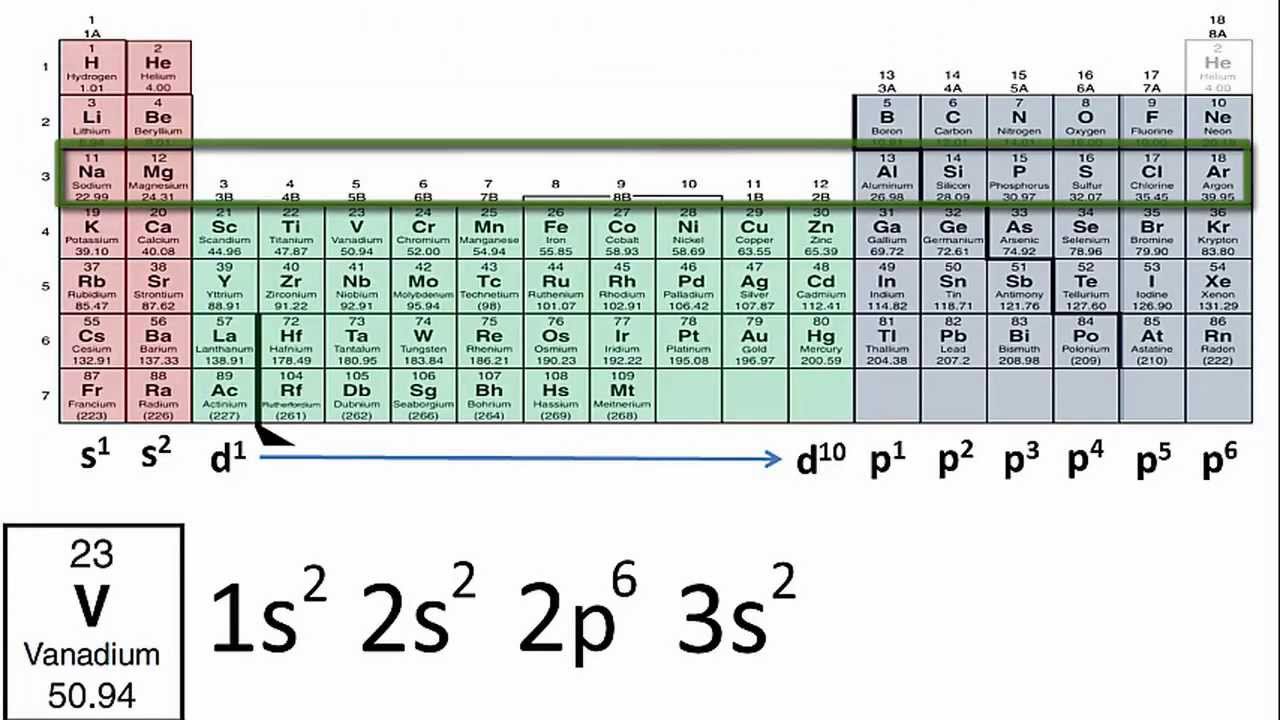
How to write valence electron configuration. So, the remaining four electrons will enter the 5d orbital. For example, the electron configuration. The vanadium atom donates two electrons from the last shell to form the vanadium ion (v 2+ ).
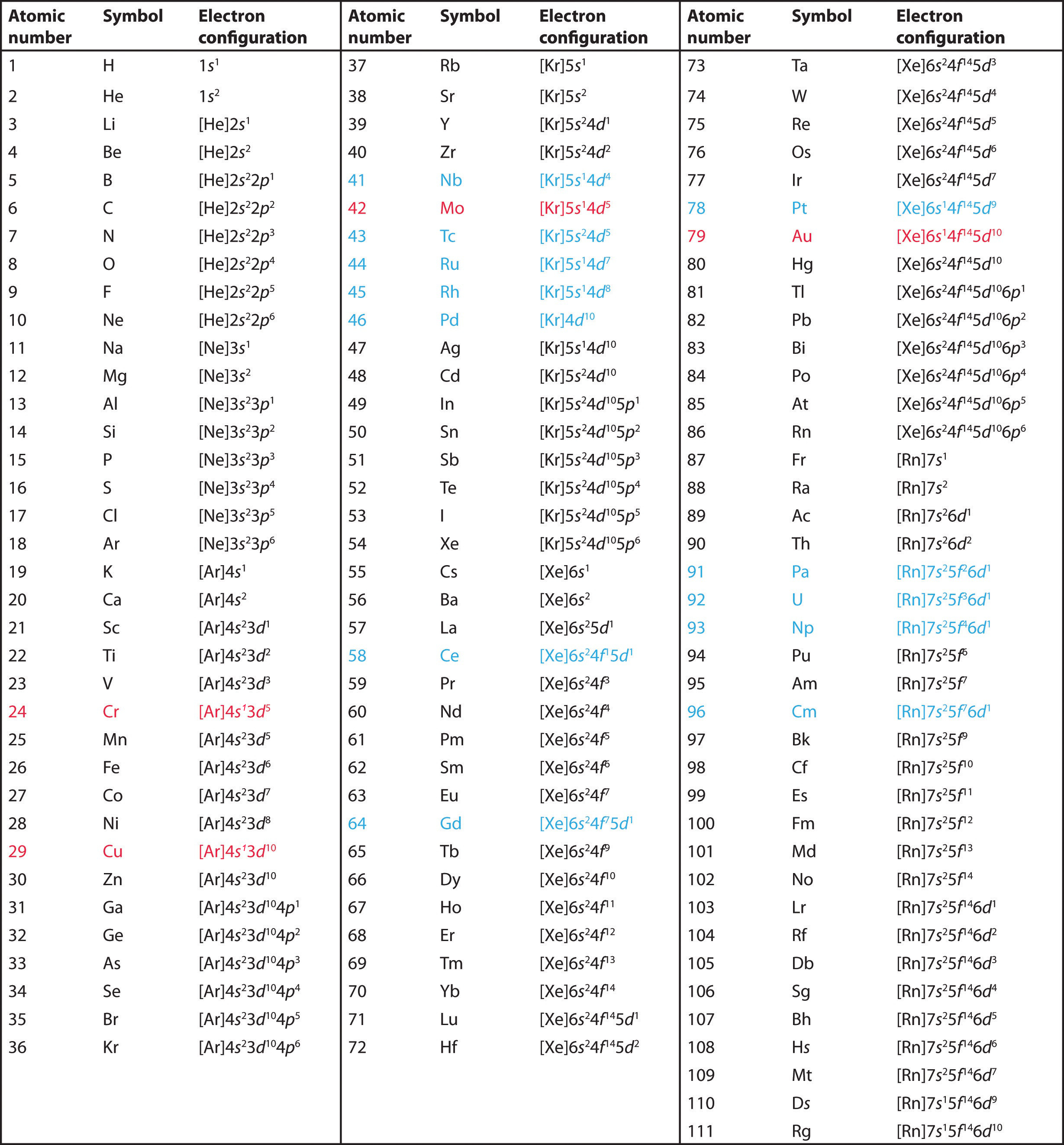
Therefore, the order of the number. In the lewis symbol for an atom, the chemical symbol of the element (as found on the periodic table) is written, and the valence electrons are represented as dots surrounding it. Therefore, the tungsten full electron configuration will be 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 4f 14 5s 2 5p 6 5d 4 6s 2.
The valency of the element is determined by. Therefore, the valence electrons of palladium are ten. Therefore, the electron configuration of bromine(br*) in an excited state will be 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p x 2 4p y 1 4p z 1 4d xy 1.
In writing the electron configuration for vanadium, the first 2 electrons will go into the 1s orbital, the next two electrons will go into the 2s orbital, and after that, the next 6 electrons will go into. Write out the electron configuration for the following elements: Palladium atom donates two electrons in the 4d orbital to convert a.
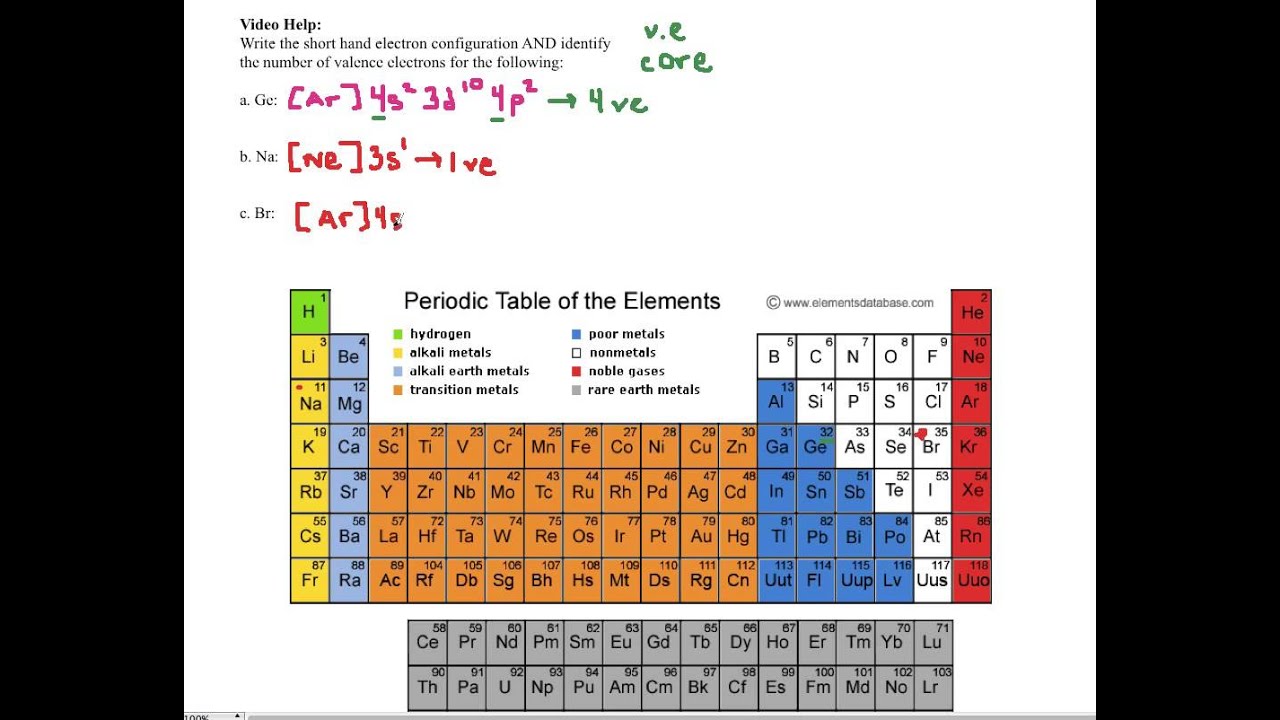
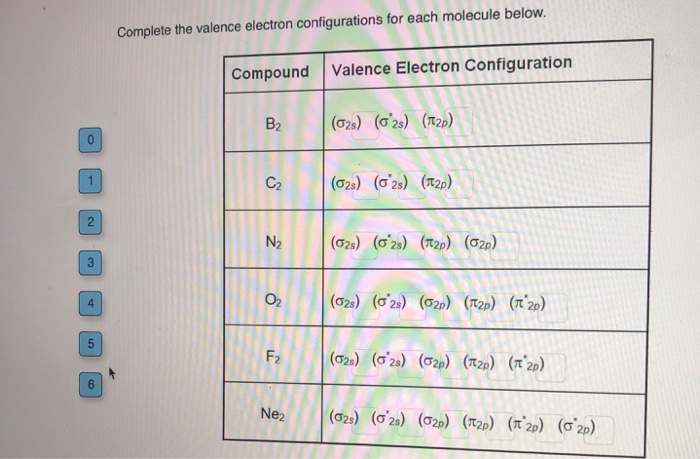
The noble gas configuration for the scandium atom is written as [ar] 4s2 3d 1. Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum. Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum.
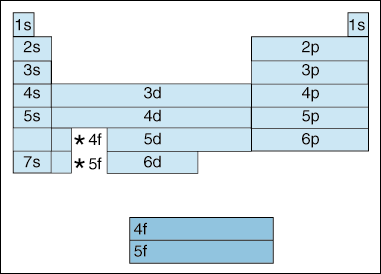
So, by this concept, we can easily find the valence electrons for the scandium atom. The valence electron configurations for all of the elements are in the periodic table below. There is a shorthand way of writing electron configurations.
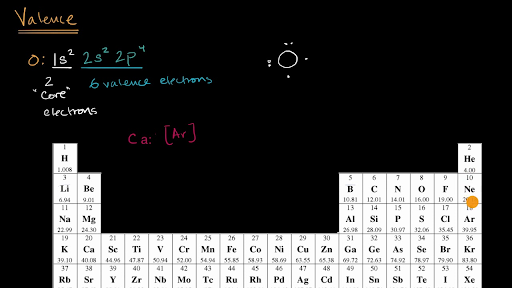
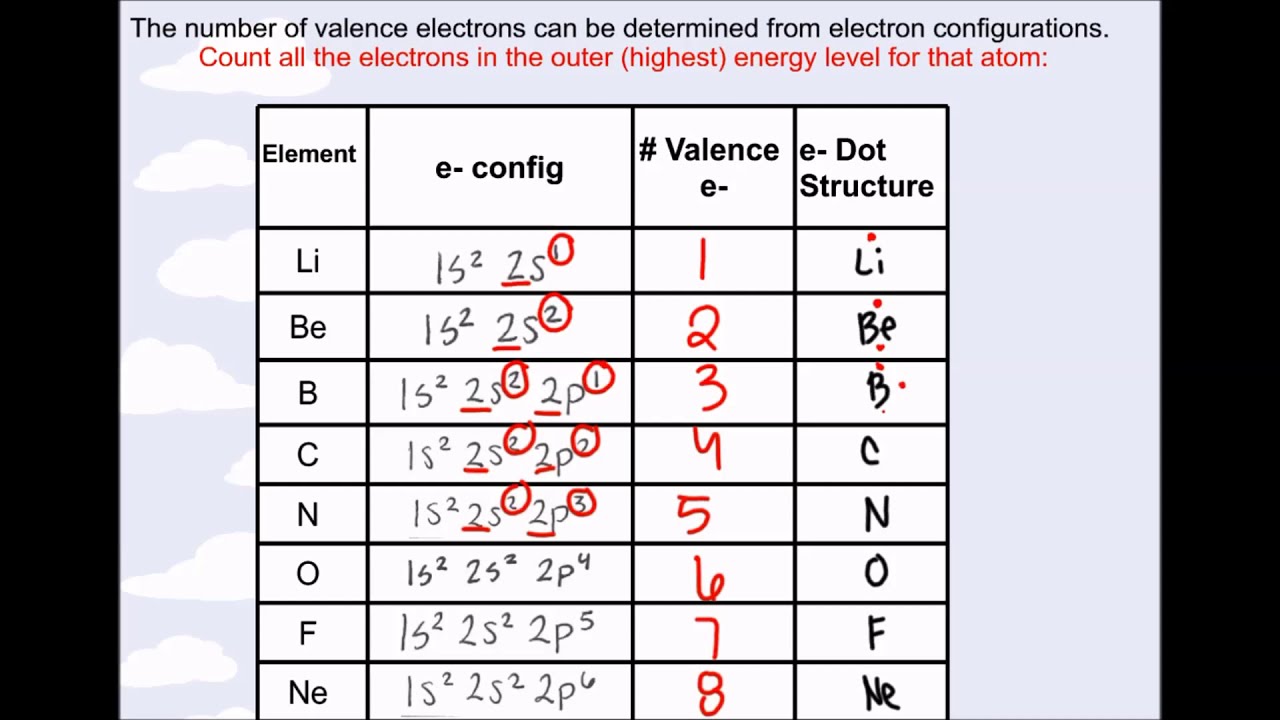
Let’s find the valence electrons of the nitrogen atom through its electron configuration.